Oramed Pharmaceuticals and the Promise of Oral Insulin
Table of contents

You know how to tell someone is an Apple user? They’ll tell you. That old joke refers to the inability for some people to keep facts hidden as they betray themselves repeatedly. We’ve been dealing with stock promoters and cheerleaders for decades now, and inevitably they’ll find their way to Nanalyze and quickly betray themselves. Even the most sophisticated of the lot (highly compensated analysts with strong subject matter expertise) quickly fall into an obvious trap. They’ll be unable to entertain any negative analysis of their sacred cow. When pushed to admit there are issues, they’ll always fall back on the classic “Tesla had the same problems too” claim. Ah yes, that old chestnut.
Companies with an easy-to-understand value proposition that have been spinning wheels for over a decade are suspicious right off the bat. It’s precisely why we’ll never invest in a company that doesn’t have meaningful revenues, with some exceptions made for companies dabbling in areas like gene editing. We were first introduced to Oramed (ORMP) by some actors on our YouTube channel who couldn’t help namedropping the stock in every other sentence. So, we decided to have a look.
About Oramed Pharmaceuticals
Originally incorporated in 2002 as a mineral exploration company, the shell that Oramed Pharmaceuticals crawled into became a pharmaceutical company “engaged in the development of innovative pharmacological solutions” starting in 2006 with their first 10-K filed in 2012. In an obvious conflict of interest, the CEO’s own mother also acts as the Chief Scientific Officer of the company whose research has led to a drug delivery method that is said to enable oral delivery of insulin for diabetes patients.
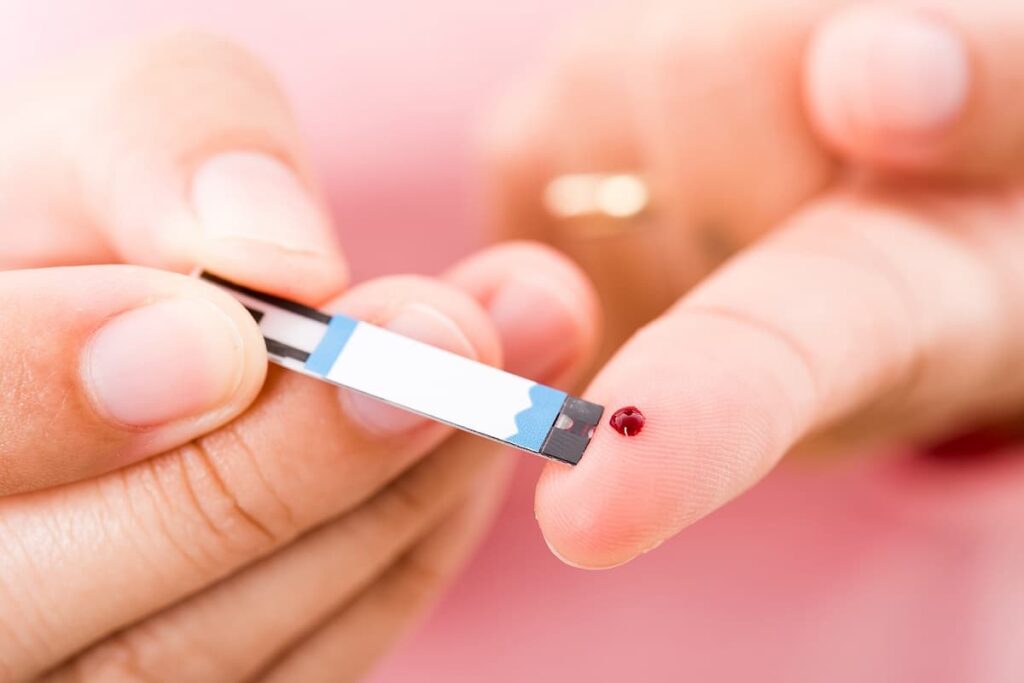
Insulin pills have been in development for decades, but one thing holding them back has been low levels of absorption — too much of the insulin in the pills breaks down in the stomach.
Credit: Freelink
Oramed claims to have developed a viable way to deliver sufficient amounts of insulin in pill form – their flagship therapeutic, ORMD-0801. It’s a bold claim because the world’s biggest insulin maker – Novo Nordisk – wasn’t able to make it happen and recently changed directions to another therapeutic – RYBELSUS – that’s currently being plastered all over USA television right now. Then there’s Novo’s once-weekly basal insulin therapy which just wrapped up Phase 3 trials and will soon be seeking FDA approval. That therapy is slightly ahead of Eli Lilly’s once-a-week insulin shot which is currently in Phase 3 trials.
The path to freedom from insulin shots is paved with failures. Pfizer spent $2.8 billion developing a novel insulin inhaler which they pulled from the market in 2007. With three companies producing 90% of the world’s insulin (Novo Nordisk, Sanofi, and Eli Lilly), you can be sure all delivery methods have been vetted, and the most viable are receiving R&D funding. Oramed has been running clinical studies for over a decade now, and they’re beginning to sound like a broken record.
Clinical Studies Galore
Whenever we analyze a stock that doesn’t have meaningful revenues, we’ll start researching as far back as possible. For Oramed, our story starts a decade ago when they engaged a clinical research organization, Medpace, to help with clinical trials.
In September 2012, we entered into a Master Service Agreement with Medpace, Inc., or Medpace, to retain Medpace as a clinical research organization for our upcoming Phase 2 clinical trial for an oral insulin capsule that is expected to start in the first calendar quarter of 2013 in the United States, and is expected to be completed in September 2013.
The following year, the 2013 10-K discussed “communicating with the FDA” regarding the IND application, and, “conducting a Phase 2a substudy before we may proceed with the Phase 2b clinical trial” which they expected to complete in Q4-2013 with the Phase 2b clinical trial to begin in Q3-2014. By 2016 they had completed “a Phase IIb clinical trial on 180 type 2 diabetic patients that was conducted in 33 sites in the United States” and went on to initiate “an additional Phase IIa dose finding clinical trial” to determine the best dosage to administer to patients. In 2017, the FDA advised that the regulatory pathway for the submission of ORMD-0801 would be a Biologics License Application, or BLA. For whatever reason, Oramed still hasn’t done that. Instead of filing for a BLA in 2017, Oramed did what they do best. They filed more studies.
- In June 2018, we initiated a glucose clamp study which will quantify insulin absorption in type 1 diabetic patients treated with ORMD-0801.
- In June 2018, we also initiated a food effect trial in the United States for ORMD-0801.
- In October 2018, we initiated an exploratory clinical study of ORMD-0801, in patients with nonalcoholic steatohepatitis, or NASH.
Fast forward to today and they’re still talking about the same trials, the same status updates, with still no BLA application filed. Here are two company statements from an article in BioPharma-Reporter.com this past February.
- Our current clinical focus is on t2D for oral insulin and we are targeting the end of next year to file a BLA application
- In addition, the company expects to complete enrollment for its oral insulin NASH trial with initial data this year.
If you’re tempted to think that Oramed has done nothing but conduct studies without providing any sort of timeline to commercialization, you’d be right. Other pundits took note of this as far back as 2014, one being the Observer which coined a piece titled Tough to Swallow: Paper Trail Behind ‘Breakthrough’ Leads to Penny-stock Profiteers. It’s easy enough to find disgruntled journalists out there, so we did some poking around ourselves. Here’s what we found.

Questionable Characters
A February 2014 investor deck notes Oramed coverage from a firm called Aegis Capital Corp. which has a checkered past, to say the least. In 2015 they were charged with “improperly selling unregistered penny stocks and for related supervisory failures,” and fined $950,000 by FINRA. Their latest stunt – making “unsuitable recommendations of highly complex variable interest rate structured products” – landed them in hot water this year with the SEC.
(Editors note: Americans, maybe you should focus less on petty political squabbling and more on what pitfalls exist out there for your elderly relatives to piss your inheritances away on.)
If were involved in the Aegis Capital Corp. hot mess, you’d want to move on as quick as possible. That’s precisely what one of their analysts did as he’s being sued alongside his colleagues for their actions with another pharmaceutical firm. Incredibly, he’s still peddling Oramed Pharmaceuticals and – as of July 2022 – was issuing a price target of (checks notes) $32 a share.

As if this information wasn’t enough to make the stock about as appealing as leprosy, there are many more red flags being thrown off that don’t need much research to uncover.
Red Flags Galore
After spending decades researching failed companies borne from the penny stock shell company world, it’s predictably easy to uncover red flags once you start doing due diligence because you know all the places to look. Here are just some red flags being waved by Oramed Pharmaceuticals (red flags taken from our piece on Investing in Penny Stocks for Dummies with our comments in italics):
- Borne from a shell company and graduated to a notable exchange via an OTC exchange From mineral exploration to drug delivery
- The company possesses one patent or a small number of patents that it claims can cure cancer and the common cold all at once. Proprietary drug delivery technology
- These patents are usually acquired in the reverse merger or invented by someone in senior management whose credentials and past successes are heavily touted. The CEO’s mom.
- Many individuals on stock message boards such as the ones found on Investors Hub will suddenly start posting messages touting the merits of the stock using phrases such as “load up”, “get on the train”, “the next Microsoft”, “easy ten bagger”, etc. Yep
- The company will keep announcing partnerships, memorandums of understanding (the legal equivalent of a handshake), and other types of agreements that never actually go anywhere. More clinical trials, anyone?
- The company will pay investor relations firms for coverage Check
- When the original opportunity does not transpire, the company will then “pivot” into other application opportunities for their technology, often latching on to whatever happens to be dominating news headlines. COVID vaccine pills, anyone?

The red flags we’ve highlighted today are the tip of the iceberg for Oramed Pharmaceuticals, a company we’ve already wasted enough time and energy looking at.
Conclusion
Where there is smoke, there’s fire. When you start to uncover red flags in a small cap firm that’s been spinning wheels for over a decade, you turn and walk away. When the world’s biggest producer of insulin throws in the towel on insulin pills after spending a whole lot more on research than Oramed did, it’s time to move on to something bigger and better. While researching this stock, we saw nearly every red flag across the board except the cheerleaders coming around and accusing us of being short. In the comments section below that’s where they’ll start to show up. You know how to tell someone’s a cheerleader? It’s the same way you’ll know someone is an Apple user.
Sign up to our newsletter to get more of our great research delivered straight to your inbox!
Nanalyze Weekly includes useful insights written by our team of underpaid MBAs, research on new disruptive technology stocks flying under the radar, and summaries of our recent research. Always 100% free.














Thanks for these articles. I’m learning a lot about importance of red flags.
Btw. Nobody pumping the stock here! Does it mean this stock could be next Microsoft? 😀
This is the next Microsoft AND the next Tesla all rolled into one. 😉 Glad you’re learning from these pieces as we’ve seen these same stories played out time and time again.
Yep, the cheerleaders came around with their false accusations exactly as we predicted! Our video on YouTube and the tweet we sent out sharing this article both attracted accusations of being short from dubious actors. Will these people never learn?